Cancer & Glucose Metabolism
Does Sugar Feed Cancer?
Most Cancers Use Glucose
Different kinds of cancers seem to have different preferences for sugars, amino acids that make protein, and fats. But most cancers use glucose, a form of sugar.

Heather Christofk, PhD, co-leader of the UCLA Metabolomics Center and a member of the Jonsson Comprehensive Cancer Center and the Broad Stem Cell Research Center, is working to identify the proteins within cancer cells that are responsible for altered glucose metabolism.
How cells break down glucose
When glucose enters a cell, the first pathway it encounters is glycolysis – the process that breaks down sugar to extract energy. Glycolysis transforms glucose into products that can produce more energy for cells. These products include ATP, cells’ energy source, and pyruvate, a versatile biological molecule that is very important to all living cells.
The body can continue breaking down the resulting products into several types of compounds that cells need:
- Amino acids, which create protein
- Fatty acids that make lipids
- Nucleic acids that make DNA and RNA, the building blocks of genes
Viruses as a model for cancer metabolism
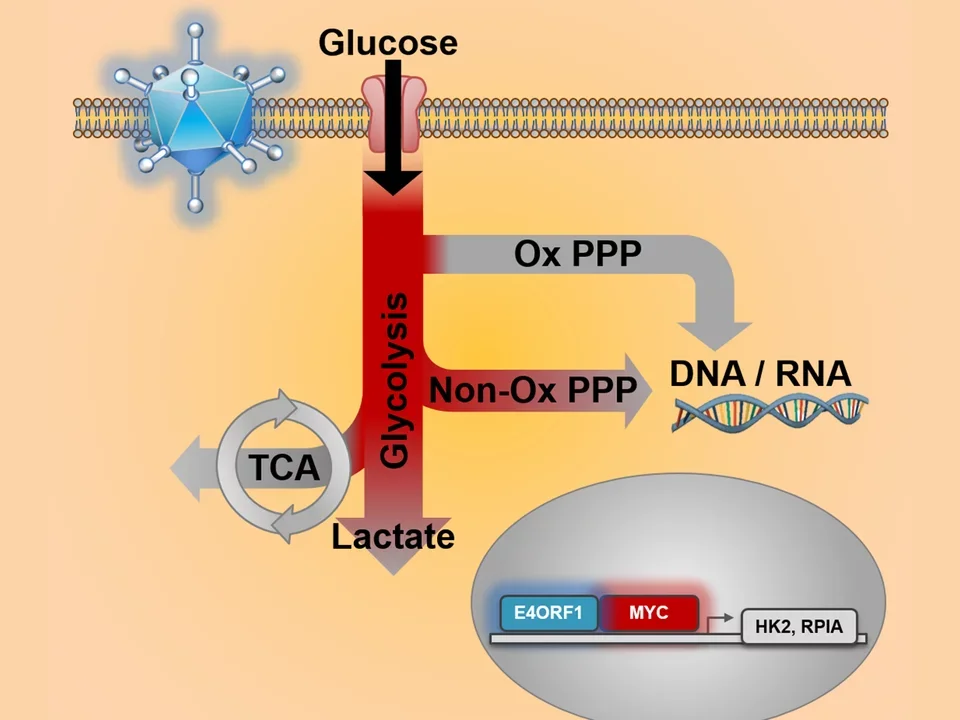
Adenovirus Model
To help find out how cancers ramp up glucose metabolism, Christofk, an associate professor of molecular and medical pharmacology and biological chemistry, is studying another life form that needs extra cellular energy to quickly and elegantly produce more copies of itself – a virus.
Christofk reasons that, because both a virus and a cancer use the same mechanism to ramp up energy production, decoding one would reveal both.
In 2014, in the journal Cell Metabolism, Christofk and her team published the first study explaining in detail how viruses reprogram the metabolism of human lung cells to promote continued viral growth. These molecular mechanisms had been unknown before her study.
In that study, the scientists learned how an adenovirus (responsible for the common cold) reprograms a human cell to take in and process more glucose. Find out more about Christofk’s work with the UCLA Metabolomics Center.
Relationships between viral and tumor metabolism
“So far, we have seen that the key metabolic nodes that are turned on in the case of viral infection are also important for cancer proliferation. The difference is that viruses are very efficient. They only turn on exactly what needs to be turned on to efficiently reprogram cell metabolism to build more virus,” Christofk says. “Viruses have figured out which step is the key step, while cancers have not. Cancers turn many things on that may not need to be activated.”
Recent experiments have shown that when key viral metabolic “nodes” are inhibited, the virus cannot replicate. Inhibiting those same pathways in cancer slows tumor growth.
Christofk is now studying the use of other nutrients critical to viral and tumor metabolism. UCLA researchers also are studying the glucose pathway from the opposite direction.
“It is fascinating to think that a drug that shuts down viral replication may also stop cancer growth.”