Astrocytes
Astrocytes Function
What Are Astrocytes?
Human brains are comprised of neurons, glia, and vascular cells. Neurons were believed to be the only cell type involved in information processing and therefore have been the focus of neuroscience research. The development, physiology, and function of glia and vascular cells remain poorly understood. In recently years, scientists made one surprising finding after another demonstrating that glia are essential for brain development and function. For example, the most numerous type of glia, astrocytes (or star-shaped cells from Greek), induces formation of synapses, which are the tiny connections through which information flows from one neuron to another within neural circuits.
Astrocytes have been implicated in a range of neurological and psychiatric disorders, including Alzheimer’s disease, Parkinson’s disease, epilepsy, stroke, traumatic brain injury and autism. Certain aspect of astrocyte function helps neural repair whereas astrocyte dysfunction exacerbates diseases. Scientists are only beginning to understand what roles astrocytes play in each type of brain disorder.
To systematically study the function of neuron, glia, and vascular cells, Dr. Ye Zhang at UCLA and her colleagues in Dr. Ben Barres’s laboratory at Stanford University found antibodies that target specific types of glia, neuron, and vascular cells in human and mouse brains and used these antibodies to isolate each cell type. They then characterized the transcriptome profile of each cell type, which is a catalog of all the proteins a cell type can make. This catalog provides a roadmap for understanding protein function in neuron, glia, and vascular cells in the brain. For example, when scientists discover that a certain protein is responsible for a neurological disorder, they can look up the new catalog to find out which cell type(s) makes the protein.
The information would help scientists quickly focus their effort on the cell type(s) relevant for the disease. Many genes have been shown to increase risk of autism. Scientists have already been studying the function of these genes in neurons. Dr. Zhang and Barres’s catalog showed that many autism-associated genes are actually enriched in glia, suggesting that glia dysfunction may contribute to autism.
Although laboratory mouse models are cornerstones of neuroscience research, about 90% of drug candidates for neurological disorders that succeed in mouse models fail in human clinical trials.
Human Astrocytes

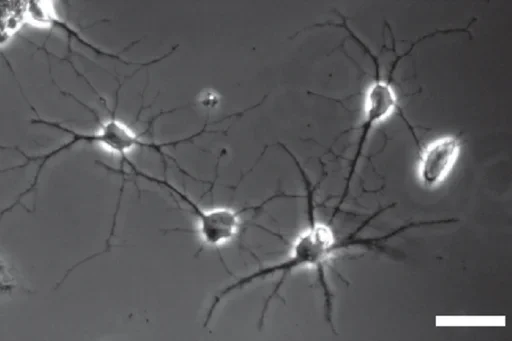
Understanding human-mouse differences is critical for translating research discoveries to new treatments in humans. Dr. Zhang and colleagues developed a new method to isolate astrocytes from surgically resected human brain tissue and to grow them in a dish. These human astrocytes in a dish showed similar function as mouse astrocytes in promoting neuron survival, synapse formation and function, and synapse elimination. However, human astrocytes make hundreds of proteins that mouse astrocytes don’t. The new astrocyte isolation and culture method enables scientists to study changes of astrocytes in human brain disorders and to test drugs directly in human cells. Currently, Dr. Zhang’s group is investigating what signals in developing brains make astrocytes mature and stop them from dividing. She hopes that identifying the signals could reveal new approaches to treating brain tumors, which resemble perpetually dividing immature glia.