Underlying Causes of Brain Disorders
Merging Human Genetic Findings and Clinical Observations

Millions of people are affected by brain disorders ranging from age-dependent neurodegenerative disorders such as Alzheimer’s, Parkinson’s and Huntington’s disease to psychiatric disorders such as Schizophrenia, depression, and autism spectrum disorders. Rapid advances in two areas of biology provide new hope to better understand these complex disorders. First, advances in human genetics and genome sequencing have greatly accelerated the discovery of genetic mutations that either cause or enhance the risk for these disorders.
Second, new tools to study the brain have vastly expanded our knowledge of how the brain controls behavior. However, translating such knowledge into new therapies to cure these diseases has been very slow. To bridge this gap, we need a fundamental understanding of the molecular and cellular networks that normally regulate brain function, and a precise knowledge of the genes that may cause these diseases. Such transformative advances in neuroscience are dependent on seamless collaboration among investigators from multiple disciplines such as biology, computer science and engineering.
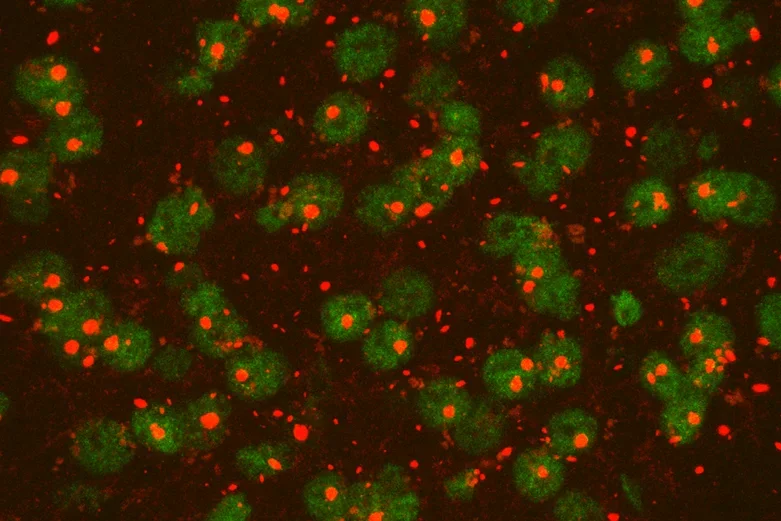
Mutant huntingtin, the causal disease protein in Huntington's disease, forms pathological protein aggregates in a mouse model of HD.
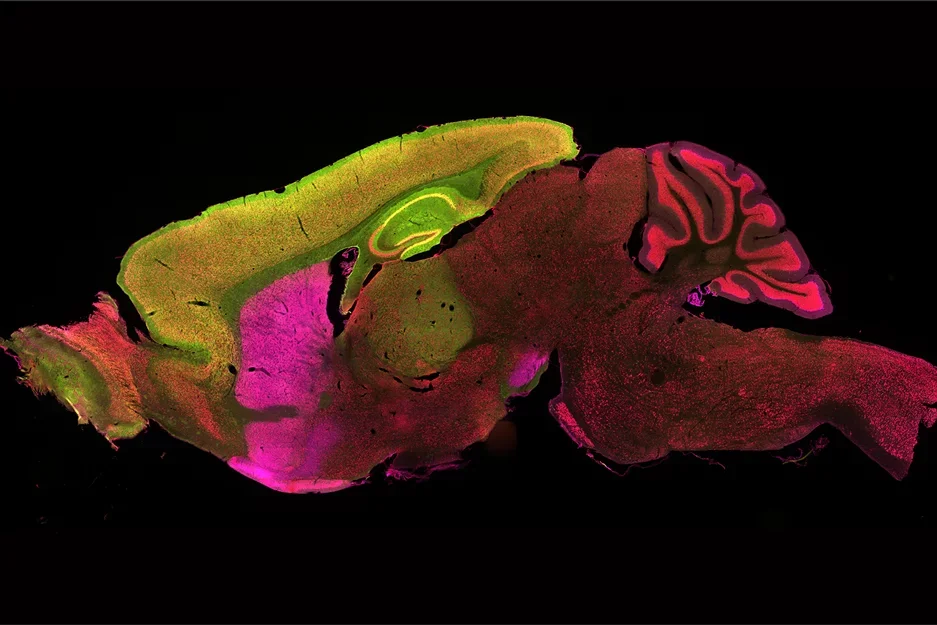
The Yang Lab showed two distinct brain regions, the cortex and striatum (labeled yellow and purple respectively), have distinct and synergistic contribution to pathogenesis of Huntington's disease.
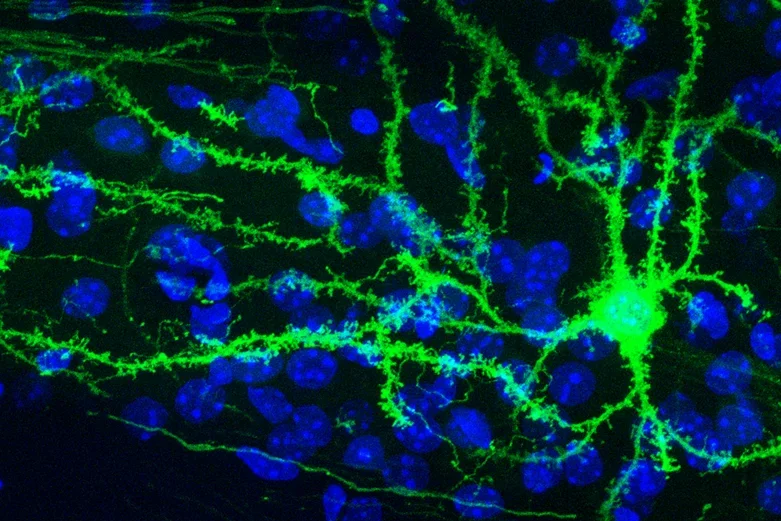
A single medium spiny neuron in the mouse striatum labeled using genetically directed Mosaicism with Repeat Frameshift (MORF) cell labeling technology.
The Science
UCLA Neuroscientist X. William Yang, MD, PhD, merges human genetic findings and clinical observations from patients with cutting-edge genetic tools to study a variety of brain disorders, including Huntington’s, Parkinson’s, and Alzheimer’s diseases. His laboratory pioneered new genetic tools to develop models that mimic neurodegenerative disorders. He used these models to study disease mechanisms and test drug targets that could lead to future treatments for these devastating disorders.
His laboratory also studies a group of brain circuits called the basal ganglia that are involved in motor performance, motor skill learning, and reinforcement learning. Their innovative research on this circuitry provides new insights into the behavioral impairment seen in opiate addiction and Tourette Syndrome.
Finally, Dr. Yang’s team is collaborating with researchers around the globe to map out the detailed molecular networks that go awry in Huntington’s disease, and to develop an unbiased pipeline to identify potential targets for future treatments.
The Discoveries
- Building innovative models to define and test drug targets for neurodegenerative diseases. Dr. Yang’s lab developed new genetic models of Huntington’s disease. He used these models to identify potential drug targets for Huntington’s disease, including a small region of the Huntingtin protein that acts as a switch to turn the disease on and off. Importantly, Dr. Yang has translated these innovative approaches to study other neurodegenerative diseases such as Alzheimer’s and Parkinson’s.
- Identifying drug targets by linking genetic data from clinical and laboratory studies. Dr. Yang co-leads a diverse group of scientists from universities, foundations and companies around the world. Their goal is to combine data from patients in the clinic with data from laboratory models of Huntington’s disease. This innovative approach has created a map of the complex molecular networks that are affected in this disease. This map will generate a list of candidate targets that will be tested by Dr. Yang’s group.
- Creating new tools to study brain disorders. Dr. Yang’s lab has developed a new set of tools to study changes that occur during behaviors such as drug addiction. One method will examine how different types of neurons express different genes. A second method will sparsely label neurons in order to study how the brain, and specifically the basal ganglia, develops and normally functions. These same tools can be used to monitor and visualize early stages of neurodegeneration. This knowledge will be crucial for early disease intervention.