Cardiovascular Research
The future of cardiovascular research and medicine is here at UCLA.
We have a team of cardiovascular research scientists who are fully committed to clinical care, discovery science and engineering, community outreach and education.
Investments of all sizes empower transformations.

Theme Spotlights
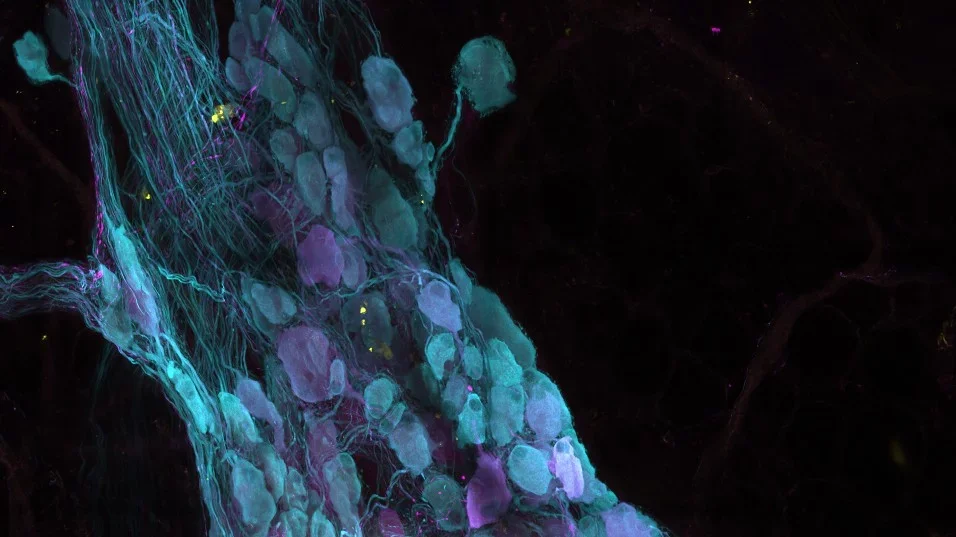
Julia Mack, PhD & Soon-Gook Hong, PhD
While laminar blood flow is known to promote endothelial resilience, the specific signaling mechanisms that connect laminar flow to vascular protection have remained elusive. This study, published in the Journal of Clinical Investigation (JCI), describes a signaling mechanism in endothelial cells that protects against inflammation. Soon-Gook Hong, postdoc in the Mack Lab, discovered that laminar flow-induced endothelial cell body elongation establishes polarized mechanosensitive signaling domains. These sub-cellular domains at the downstream end of the cell facilitate focal calcium entry via TRPV4 channels to produce nitric oxide. Silencing domain activity results in spontaneous inflammation and activating TRPV4 channels limits inflammation during exposure to an inflammatory agonist. “We hope this work, which defines a mechanosensitive signaling feature in the endothelium, will have therapeutic potential to protect against or treat vascular diseases,” said Julia Mack, Assistant Adjunct Professor in the Division of Cardiology, and the corresponding author. This work was funded in part by an award from the UCLA Cardiovascular Discovery Fund.
Yijie Wang, PhD
A new study published in Science and led by Yijie Wang, a postdoctoral fellow in Deb Laboratory, and Dr. Deb Arjun, senior author, professor of medicine and director of the UCLA Cardiovascular Theme at the David Geffen School of Medicine, is promising for those with irregular heartbeats, or arrhythmias. The researchers demonstrated that heart arrhythmias are promoted by fibroblasts, scar-forming cells found in scar tissues of the injured heart. Newly identified mechanisms of fibroblast-myocyte communication have clinical implications and these findings may be broadly applicable to treatment of the nervous system.
Jake Lusis, PhD
In this recent study published in Science, postdoctoral researcher Yang Cao, Professor Jake Lusis, and colleagues share findings about liver-heart communication via a liver-derived protein, coagulation factor XI (FXI), and the role that it plays in heart injury.
Peter Hanna, MD
Peter is currently a cardiology fellow and PhD candidate in Molecular, Cellular and Integrative Physiology at UCLA. As part of the Specialty Training and Advanced Research (STAR) program, he used a multiscale approach to create a comprehensive atlas of how the cardiac autonomic nervous system controls the heartbeat.
Jeffrey Hsu, MD, PhD
While vascular calcification is associated with a higher risk of cardiac events, whether modifying calcification improves outcomes is unclear. New studies show that using fused micro-PET/CT imaging in mice allows researchers to better understand how cardiovascular calcification responds to interventions, such as endurance exercise and parathyroid hormone therapy.
Faculty News: NIH Director’s Transformative Research Award
Backed by a $6.2 million grant from the National Institutes of Health, UCLA Cardiovascular Research Theme members will lead a study of how COVID-19 causes multiple organ failure.
Vascular Biology
Leveraging a UCLA Cardiovascular Discovery Fund Award to study calcium signaling.
Cardiac Injury
Developing displacement analysis of myocardial mechanical deformation (DIAMOND.)
Primary Endocardial Fibroelastosis (pEFE)
A recent study by UCLA investigators solves the genetic diagnosis of a devastating form of congenital heart disease.